Autopsies of two teenage boys who died days after receiving Pfizer’s COVID vaccine prove the vaccine caused their deaths. Pathological findings suggest there may be a way to distinguish SARS-COV-2 infection-induced myo/pericarditis from vaccine-induced cardiac injury. Vaccine-induced heart injury can be sub-clinical, but how often?
Pathologists who examined the autopsies of two teenage boys who died days after receiving Pfizer’s COVID-19 vaccine concluded the vaccine caused the teens’ deaths.
The three pathologists, two of whom are medical examiners, published their findings Feb. 14 in an early online release article, “Autopsy Histopathologic Cardiac Findings in Two Adolescents Following the Second COVID-19 Vaccine Dose,” in the Archives of Pathology and Laboratory Medicine.
The authors’ findings were conclusive. Two teenage boys were pronounced dead in their homes three and four days after receiving the second Pfizer-BioNTech COVID-19 dose.
There was no evidence of active or previous COVID-19 infection. The teens had negative toxicology screens (i.e. no drugs or poisons were present in their bodies).
These boys died from the vaccine.
Histopathological examination of their cardiac tissue revealed an important new finding: Neither heart demonstrated evidence of typical myocarditis.
Instead, the authors found evidence of microscopic changes consistent with a different form of heart injury called toxic cardiomyopathy. They wrote:
“The myocardial injury seen in these post-vaccine hearts is different from typical myocarditis and has an appearance most closely resembling a catecholamine-mediated stress (toxic) cardiomyopathy.”
The authors further explained what they observed under the microscope:
“Their histopathology does not demonstrate a typical myocarditis … In these two post-vaccination instances, there are areas of contraction bands and hypereosinophilic myocytes distinct from the inflammation.
“This injury pattern is instead similar to what is seen in the myocardium of patients who are clinically diagnosed with Takotsubo, toxic or ‘stress’ cardiomyopathy, which is a temporary myocardial injury that can develop in patients with extreme physical, chemical, or sometimes emotional stressors.
“Stress cardiomyopathy is a catecholamine-mediated ischemic process seen in high catecholamine states in the absence of coronary artery disease or spasm. It has also been called ‘neurogenic myocardial injury’ and ‘broken heart syndrome.’”
The pathologists determined there was a different mechanism of heart injury at play in these two boys, distinct from a purely infectious process that would result directly from a viral infection like COVID-19.
This is an important finding. There may be a way to distinguish cardiac injury resulting from a SARS-COV-2 infection from cardiac injury where the vaccine predisposes the patient to stress cardiomyopathy before contracting COVID-19.
However, the authors are careful not to assume that cardiac injuries from COVID-19 and COVID-19 vaccines can always be sorted out under the microscope.
They explain that stress cardiomyopathy, or “broken heart syndrome,” may also occur in a rare hyperinflammatory state that is known to occur in COVID-19 infection as well:
“This post-vaccine reaction may represent an overly exuberant immune response and the myocardial injury is mediated by similar immune mechanisms as described with SARS-COV-2 and multisystem inflammatory syndrome (MIS-C) cytokine storms.”
The authors admit this pathological finding may also occur as a result of MIS-C, a known complication of SARS-COV2 infection.
Learning more about this condition requires a biopsy of heart tissue, or in this case an autopsy. We know very little about the nature of myocarditis in people who are clinically stable because heart biopsies are not conducted on them and autopsies are rarely done on patients who die from COVID-19.
There still is no practical way of screening for cardiac injury beyond assessing symptoms.
Unfortunately, the two boys did not have symptoms of myocarditis (fever, chest pain, palpitations, or dyspnea) prior to their cardiac arrest and death. One complained of a headache and gastric upset which resolved. The other had no complaints.
This is extremely concerning. These boys had smoldering, catastrophic heart injuries with no symptoms.
How many others have insidious cardiac involvement from vaccination that won’t manifest until they get a serious case of COVID-19 or the flu? Or perhaps when they subject themselves to the physical stress of competitive sports?
These findings suggest a significant subset of COVID-19 deaths in the vaccinated could be due to the vaccines themselves.
Furthermore, it raises this question: How often does this condition exist in a latent form in vaccinated individuals?
The CDC believes the risk of vaccine-induced myocarditis not significant
The Centers for Disease Control and Prevention (CDC) says the risk of myocarditis and pericarditis in adolescents who get the COVID-19 vaccine is “extremely rare” and “most cases are mild.”
But those assurances conflict with the agency’s own data.
The CDC’s Advisory Committee on Immunization Practices (ACIP) presented this disquieting information (see chart below) during its June 23, 2021 meeting convened specifically to address the risks of myo/pericarditis in 12- to 15-year-olds who received Pfizer’s COVID vaccine:
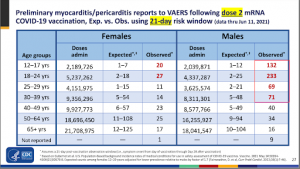
This slide is important for two reasons.
First, the incidence of this potentially lethal condition is significantly higher in the vaccinated (“Observed” column) compared to the background rate (“Expected” column), especially in males in the 18- to 24-year-old age range.
In the 12- to 17-year-old male cohort, the risk of myo/pericarditis is at least 11 times higher than the background rate.
With more than 2 million doses administered at the time when these cases of myo/pericarditis were identified, we can be confident these data represent an undeniable safety signal.
The second reason this slide is important is this: The CDC is drawing directly from the Vaccine Adverse Event Reporting System (VAERS), a system specifically designed to monitor for safety signals when vaccines are administered to the public.
As of Feb. 15, the CDC continues to assure the public that “Reports of adverse events to VAERS following vaccination, including deaths, do not necessarily mean that a vaccine caused a health problem.”
In essence, the CDC is acknowledging that reports of deaths and other adverse events following vaccination exist in VAERS but do not comprise any risk because causality has not been verified.
Then why did the ACIP choose to accept VAERS as a legitimate source of information on myo/pericarditis in their calculations?
The CDC released its conclusions immediately following the ACIP meeting:
“The facts are clear: this is an extremely rare side effect, and only an exceedingly small number of people will experience it after vaccination. Importantly, for the young people who do, most cases are mild, and individuals recover often on their own or with minimal treatment.”
But how do they know this?
One month after this comforting statement from the CDC, the U.S. Food and Drug Administration (FDA) admitted in this letter to Pfizer that the agency was not able to adequately assess the risk of myocarditis from Pfizer’s product:
“We have determined that an analysis of spontaneous postmarketing adverse events reported under section 505(k)(1) of the FDCA [Federal Food, Drug and Cosmetic Act] will not be sufficient to assess known serious risks of myocarditis and pericarditis and identify an unexpected serious risk of subclinical myocarditis.
“Furthermore, the pharmacovigilance system that FDA is required to maintain under section 505(k)(3) of the FDCA is not sufficient to assess these serious risks.”
Commenting on the FDA’s letter, Dr. Meryl Nass said, “The FDA is saying that neither an analysis of the data in VAERS or of any of the other taxpayer-funded databases will provide sufficient assessment of the risk of this product.”
“This is a joke,” said Nass, adding:
“All this data, plus software, plus a team of analysts, and the FDA says it can’t assess the risk of myocarditis, despite identifying thousands of cases?
“Furthermore, unsaid, but implied by the FDA, is that if the FDA is incapable of assessing the risk of myocarditis despite thousands of reported cases, it cannot or will not be capable of assessing the other serious adverse events that have been reported in conjunction with COVID vaccines.”
If the FDA is not able to perform adequate surveillance of safety signals around vaccine-induced myocarditis, who will?
The FDA assigns this unenviable but essential task to Pfizer itself (again, from the FDA’S letter to Pfizer):
“Therefore, based on appropriate scientific data, we have determined that you are required to conduct the following studies…”
Is myocarditis ‘extremely rare’ after COVID-19 vaccination?
As of Feb. 4, VAERS reported 495 cases of myo/pericarditis in 12- to 17-year-olds. VAERS data show that as of Feb. 10, there were 2,239 reported cases of myocarditis in people under the age of 30.
However, a widely cited CDC-sponsored study (Lazarus et al) concluded the incidence of adverse events is 10 to 100 times higher than are reported to VAERS.
More recent calculations estimate that adverse events are underreported by a factor of approximately 41.
From these estimates, we can conclude there may have been approximately 20,000 cases of myocarditis in 12- to 17-year-olds since Pfizer’s COVID-19 vaccine received Emergency Use Authorization and was rolled out to this age group..
The VAERS data from June 11, 2021 from the table above show 132 cases of myo/pericarditis were observed in 2,039,000 doses given to 12- to 17-year-old males. This is approximately 6.5 cases in 100,000 doses.
This study from Hong Kong found the incidence of myo/pericarditis after two doses with Pfizer’s Comirnaty vaccine was 37 in 100,000. This incidence matches nearly exactly with findings from this study that used the Vaccine Safety DataLink (VSD) system (37.7 12-17 year olds per 100,000 suffered myo/pericarditis after their second dose). This is more evidence that significant underreporting is in play in the VAERS system.
Will most of these teens “recover on their own”? How many other vaccinated people have varying degrees of “broken heart” syndrome that remain asymptomatic, undiagnosed and unreported?
These new findings indicate that no one can answer these questions right now — especially not the CDC and the FDA.
If the FDA has admitted it cannot assess the risk of myocarditis using the surveillance systems in place, how then is the CDC able to assure us that the risk is low enough to continue to proceed with a vaccination campaign that now includes 5- to 11-year-old children?
The FDA has abdicated its responsibility for monitoring the safety of these vaccines to the vaccine manufacturers.
The CDC is using VAERS data in its own analyses while urging the public to discount all adverse events, including deaths, that appear in the very same database.
There isn’t any regulation happening here. Our regulatory agencies have become mouthpieces for the very industry they are tasked to oversee.
- “© 2/18/22 Children’s Health Defense, Inc. This work is reproduced and distributed with the permission of Children’s Health Defense, Inc. Want to learn more from Children’s Health Defense? Sign up for free news and updates from Robert F. Kennedy, Jr. and the Children’s Health Defense. Your donation will help to support us in our efforts.